ADVIA 2120i Hematology System
The challenging paradigm of manual slide reviews
While slide review rates can vary greatly between labs, studies show that on average, more than 20% of hematology samples trigger a manual scan or count.1,2 Reviews are critical to the release of accurate results, but high frequencies can potentially affect the laboratory in terms of both labor and cost efficiency,
leading to lower productivity (billed tests per full-time equivalent).3
Do more with less and chart a path to transform care delivery
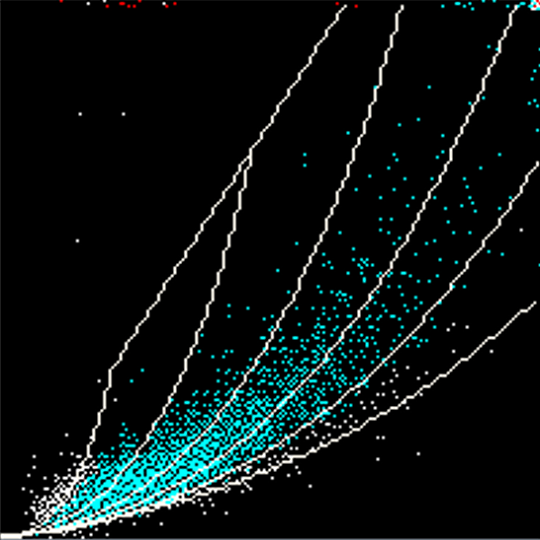
Siemens Healthineers-equipped laboratories have a lower slide review rate than their peers, despite the fact that they perform more complete blood count tests on average.*
Keele University Independent Benchmarking Study, 2017
Optimize clinical operations with technology that can show you the difference
The ADVIA® 2120i Hematology System enables high-throughput CBC, differential, reticulocyte, body fluid, and multispecies testing. Reduce ambiguity, clearly distinguish normal from abnormal, and confidently release results with innovative analytical technologies that support first-pass accuracy.
Increase workforce productivity through advanced integration
From digital morphology and data management to total lab automation, Siemens Healthineers extended portfolio can reduce time-consuming manual touchpoints across hematology testing for labs of all sizes. Integration with the ADVIA 2120i system enables you to standardize operations from start to finish, raising the bar for quality and productivity in your lab.

CellaVision® Digital Morphology Systems

Atellica® Data Manager

Aptio® Automation
Are you interested?
Share
Do you find this page interesting? Share the information with peers.
Kim, et al. Comparison study of the rates of manual peripheral blood smear review from 3 automated hematology analyzers, Unicel DxH 800, ADVIA 2120i, and XE 2100, using international consensus group guidelines. Arch Pathol Lab Med. 2012 Nov;136.
Novis D, Walsh M, Wilkinson D, St. Louis M, Ben-Ezra J. Arch Pathol Lab Med. 2006;130(5):596-601.
http://www.captodayonline.com/Archives/feature_stories/0405PeripheralBlud.html
ADVIA, Atellica, and all associated marks are trademarks of Siemens Healthcare Diagnostics Inc., or its affiliates. Aptio Automation is manufactured by Inpeco and is exclusively distributed by Siemens Healthcare Diagnostics Inc. Aptio is a trademark of Siemens Healthcare Diagnostics Inc. Inpeco is a trademark of Inpeco SA. Cellavision is a trademark of CellaVision AB. All other trademarks and brands are the property of their respective owners
The products/features mentioned here are not commercially available in all countries. Due to regulatory reasons, their future availability cannot be guaranteed. Please contact your local Siemens Healthineers organization for further details.
*The results are based on the specific settings of these labs. Because there is no typical hospital or laboratory, and many variables exist (e.g., hospital size, samples mix, case mix, level of IT and/or automation adoption), there can be no guarantee that other customers will achieve the same results. Results in other cases may vary.